Supercooling Water Bottle Before Shaking on:
[Wikipedia]
[Google]
[Amazon]

 Supercooling, also known as undercooling, is the process of lowering the temperature of a
Supercooling, also known as undercooling, is the process of lowering the temperature of a
 Constitutional supercooling, which occurs during solidification, is due to compositional solid changes, and results in cooling a liquid below the freezing point ahead of the solid–liquid
Constitutional supercooling, which occurs during solidification, is due to compositional solid changes, and results in cooling a liquid below the freezing point ahead of the solid–liquid
Supercooled liquids on arxiv.orgRadiolab podcast on supercooling
{{Authority control Thermodynamic processes Condensed matter physics Concepts in physics Glass physics

 Supercooling, also known as undercooling, is the process of lowering the temperature of a
Supercooling, also known as undercooling, is the process of lowering the temperature of a liquid
A liquid is a nearly incompressible fluid that conforms to the shape of its container but retains a (nearly) constant volume independent of pressure. As such, it is one of the four fundamental states of matter (the others being solid, gas, a ...
or a gas
Gas is one of the four fundamental states of matter (the others being solid, liquid, and plasma).
A pure gas may be made up of individual atoms (e.g. a noble gas like neon), elemental molecules made from one type of atom (e.g. oxygen), or ...
below its melting point
The melting point (or, rarely, liquefaction point) of a substance is the temperature at which it changes state from solid to liquid. At the melting point the solid and liquid phase exist in equilibrium. The melting point of a substance depends ...
without it becoming a solid
Solid is one of the State of matter#Four fundamental states, four fundamental states of matter (the others being liquid, gas, and Plasma (physics), plasma). The molecules in a solid are closely packed together and contain the least amount o ...
. It achieves this in the absence of a seed crystal or nucleus around which a crystal
A crystal or crystalline solid is a solid material whose constituents (such as atoms, molecules, or ions) are arranged in a highly ordered microscopic structure, forming a crystal lattice that extends in all directions. In addition, macro ...
structure can form. The supercooling of water can be achieved without any special techniques other than chemical demineralization, down to −48.3 °C (−55 °F). Droplets of supercooled water often exist in stratus
Stratus may refer to:
Weather
*Stratus cloud, a cloud type
**Nimbostratus cloud, a cloud type
**Stratocumulus cloud, a cloud type
**Altostratus cloud, a cloud type
**Altostratus undulatus cloud, a cloud type
**Cirrostratus cloud, a cloud type
Mus ...
and cumulus cloud
Cumulus clouds are clouds which have flat bases and are often described as "puffy", "cotton-like" or "fluffy" in appearance. Their name derives from the Latin ''cumulo-'', meaning ''heap'' or ''pile''. Cumulus clouds are low-level clouds, gener ...
s. An aircraft
An aircraft is a vehicle that is able to fly by gaining support from the air. It counters the force of gravity by using either static lift or by using the dynamic lift of an airfoil, or in a few cases the downward thrust from jet engines ...
flying through such a cloud sees an abrupt crystallization of these droplets, which can result in the formation of ice on the aircraft's wings or blockage of its instruments and probes.
Animals utilize supercooling to survive in extreme temperatures. There are many mechanisms that aid in maintaining a liquid state, such as the production of antifreeze protein
Antifreeze proteins (AFPs) or ice structuring proteins refer to a class of polypeptides produced by certain animals, plants, fungi and bacteria that permit their survival in temperatures below the freezing point of water. AFPs bind to small ...
s, which bind to ice crystals to prevent water molecules from binding and spreading the growth of ice. The winter flounder
The winter flounder (''Pseudopleuronectes americanus''), also known as the black back, is a right-eyed ("dextral") flatfish of the family Pleuronectidae. It is native to coastal waters of the western north Atlantic coast, from Labrador, Canada to ...
is one such fish that utilizes these proteins to survive in its frigid environment. In plants, cellular barriers such as lignin, suberin
Suberin, cutin and lignins are complex, higher plant epidermis and periderm cell-wall macromolecules, forming a protective barrier. Suberin, a complex polyester biopolymer, is lipophilic, and composed of long chain fatty acids called suberin aci ...
and the cuticle inhibit ice nucleators and force water into the supercooled tissue.
One commercial application of supercooling is in refrigeration
The term refrigeration refers to the process of removing heat from an enclosed space or substance for the purpose of lowering the temperature.International Dictionary of Refrigeration, http://dictionary.iifiir.org/search.phpASHRAE Terminology, ht ...
. Freezers can cool drinks to a supercooled level so that when they are opened, they form a slush
Slush, also called slush ice, is a slurry mixture of small ice crystals (e.g., snow) and liquid water.
In the natural environment, slush forms when ice or snow melts or during mixed precipitation. This often mixes with dirt and other polluta ...
. Supercooling was also successfully applied to organ preservation at Massachusetts General Hospital/Harvard Medical School
Harvard Medical School (HMS) is the graduate medical school of Harvard University and is located in the Longwood Medical Area of Boston, Massachusetts. Founded in 1782, HMS is one of the oldest medical schools in the United States and is consi ...
. Liver
The liver is a major Organ (anatomy), organ only found in vertebrates which performs many essential biological functions such as detoxification of the organism, and the Protein biosynthesis, synthesis of proteins and biochemicals necessary for ...
s that were later transplanted into recipient animals were preserved by supercooling for up to 96 hours (4 days), quadrupling the limits of what could be achieved by conventional liver preservation methods.
Explanation
A liquid crossing its standard freezing point will crystalize in the presence of a seed crystal or nucleus around which acrystal
A crystal or crystalline solid is a solid material whose constituents (such as atoms, molecules, or ions) are arranged in a highly ordered microscopic structure, forming a crystal lattice that extends in all directions. In addition, macro ...
structure can form creating a solid. Lacking any such nuclei, the liquid phase
Phase or phases may refer to:
Science
*State of matter, or phase, one of the distinct forms in which matter can exist
*Phase (matter), a region of space throughout which all physical properties are essentially uniform
* Phase space, a mathematic ...
can be maintained all the way down to the temperature at which crystal homogeneous nucleation occurs.
Homogeneous nucleation can occur above the glass transition temperature
The glass–liquid transition, or glass transition, is the gradual and reversible transition in amorphous materials (or in amorphous regions within semicrystalline materials) from a hard and relatively brittle "glassy" state into a viscous or rub ...
, but if homogeneous nucleation has not occurred above that temperature, an amorphous
In condensed matter physics and materials science, an amorphous solid (or non-crystalline solid, glassy solid) is a solid that lacks the long-range order that is characteristic of a crystal.
Etymology
The term comes from the Greek ''a'' ("wi ...
(non-crystalline) solid will form.
Water
Water (chemical formula ) is an inorganic, transparent, tasteless, odorless, and nearly colorless chemical substance, which is the main constituent of Earth's hydrosphere and the fluids of all known living organisms (in which it acts as a ...
normally freezes at 273.15 K (0 °C or 32 °F), but it can be "supercooled" at standard pressure down to its crystal homogeneous nucleation at almost 224.8 K (−48.3 °C/−55 °F). The process of supercooling requires that water be pure and free of nucleation
In thermodynamics, nucleation is the first step in the formation of either a new thermodynamic phase or structure via self-assembly or self-organization within a substance or mixture. Nucleation is typically defined to be the process that deter ...
sites, which can be achieved by processes like reverse osmosis
Reverse osmosis (RO) is a water purification process that uses a partially permeable membrane to separate ions, unwanted molecules and larger particles from drinking water. In reverse osmosis, an applied pressure is used to overcome osmotic pre ...
or chemical demineralization
A chemical substance is a form of matter having constant chemical composition and characteristic properties. Some references add that chemical substance cannot be separated into its constituent elements by physical separation methods, i.e., wit ...
, but the cooling itself does not require any specialised technique. If water is cooled at a rate on the order of 106 K/s, the crystal nucleation can be avoided and water becomes a glass
Glass is a non-crystalline, often transparent, amorphous solid that has widespread practical, technological, and decorative use in, for example, window panes, tableware, and optics. Glass is most often formed by rapid cooling (quenching) of ...
—that is, an amorphous (non-crystalline) solid. Its glass transition temperature
The glass–liquid transition, or glass transition, is the gradual and reversible transition in amorphous materials (or in amorphous regions within semicrystalline materials) from a hard and relatively brittle "glassy" state into a viscous or rub ...
is much colder and harder to determine, but studies estimate it at about 136 K (−137 °C/−215 °F).
Glassy water can be heated up to approximately 150 K (−123 °C/−189.4 °F) without nucleation occurring.
In the range of temperatures between 231 K (−42 °C/−43.6 °F) and 150 K (−123 °C/−189.4 °F), experiments find only crystal ice.
Droplets of supercooled water often exist in stratus
Stratus may refer to:
Weather
*Stratus cloud, a cloud type
**Nimbostratus cloud, a cloud type
**Stratocumulus cloud, a cloud type
**Altostratus cloud, a cloud type
**Altostratus undulatus cloud, a cloud type
**Cirrostratus cloud, a cloud type
Mus ...
and cumulus cloud
Cumulus clouds are clouds which have flat bases and are often described as "puffy", "cotton-like" or "fluffy" in appearance. Their name derives from the Latin ''cumulo-'', meaning ''heap'' or ''pile''. Cumulus clouds are low-level clouds, gener ...
s. An aircraft
An aircraft is a vehicle that is able to fly by gaining support from the air. It counters the force of gravity by using either static lift or by using the dynamic lift of an airfoil, or in a few cases the downward thrust from jet engines ...
flying through such a cloud sees an abrupt crystallization of these droplets, which can result in the formation of ice on the aircraft's wings or blockage of its instruments and probes, unless the aircraft is equipped with an appropriate de-icing
Deicing is the process of removing snow, ice or frost from a surface. Anti-icing is the application of chemicals that not only deice but also remain on a surface and continue to delay the reformation of ice for a certain period of time, or prev ...
system. Freezing rain
Freezing rain is rain maintained at temperatures below freezing by the ambient air mass that causes freezing on contact with surfaces. Unlike a mixture of rain and snow or ice pellets, freezing rain is made entirely of liquid droplets. The rain ...
is also caused by supercooled droplets.
The process opposite to supercooling, the melting of a solid above the freezing point, is much more difficult, and a solid will almost always melt at the same temperature
Temperature is a physical quantity that expresses quantitatively the perceptions of hotness and coldness. Temperature is measured with a thermometer.
Thermometers are calibrated in various temperature scales that historically have relied o ...
for a given pressure
Pressure (symbol: ''p'' or ''P'') is the force applied perpendicular to the surface of an object per unit area over which that force is distributed. Gauge pressure (also spelled ''gage'' pressure)The preferred spelling varies by country and e ...
. For this reason, it is the melting point which is usually identified, using melting point apparatus; even when the subject of a paper is "freezing-point determination", the actual methodology is "the principle of observing the disappearance rather than the formation of ice". It is possible, at a given pressure, to superheat a liquid above its boiling point
The boiling point of a substance is the temperature at which the vapor pressure of a liquid equals the pressure surrounding the liquid and the liquid changes into a vapor.
The boiling point of a liquid varies depending upon the surrounding envir ...
without it becoming gaseous.
Supercooling should not be confused with freezing-point depression
Freezing-point depression is a drop in the minimum temperature at which a substance freezes, caused when a smaller amount of another, non- volatile substance is added. Examples include adding salt into water (used in ice cream makers and for ...
. Supercooling is the cooling of a liquid below its freezing point without it becoming solid. Freezing point depression is when a solution
Solution may refer to:
* Solution (chemistry), a mixture where one substance is dissolved in another
* Solution (equation), in mathematics
** Numerical solution, in numerical analysis, approximate solutions within specified error bounds
* Soluti ...
can be cooled below the freezing point of the corresponding pure liquid due to the presence of the solute
In chemistry, a solution is a special type of homogeneous mixture composed of two or more substances. In such a mixture, a solute is a substance dissolved in another substance, known as a solvent. If the attractive forces between the solvent ...
; an example of this is the freezing point depression that occurs when salt
Salt is a mineral composed primarily of sodium chloride (NaCl), a chemical compound belonging to the larger class of salts; salt in the form of a natural crystalline mineral is known as rock salt or halite. Salt is present in vast quantitie ...
is added to pure water.
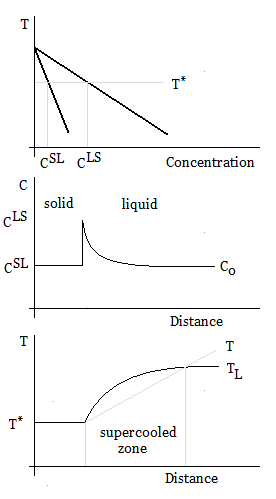
Constitutional supercooling
 Constitutional supercooling, which occurs during solidification, is due to compositional solid changes, and results in cooling a liquid below the freezing point ahead of the solid–liquid
Constitutional supercooling, which occurs during solidification, is due to compositional solid changes, and results in cooling a liquid below the freezing point ahead of the solid–liquid interface
Interface or interfacing may refer to:
Academic journals
* ''Interface'' (journal), by the Electrochemical Society
* '' Interface, Journal of Applied Linguistics'', now merged with ''ITL International Journal of Applied Linguistics''
* '' Int ...
. When solidifying a liquid, the interface is often unstable, and the velocity of the solid–liquid interface must be small in order to avoid constitutional supercooling.
Constitutional supercooling is observed when the liquidus temperature gradient at the interface (the position x=0) is larger than the imposed temperature gradient:
:
The liquidus slope from the binary phase diagram is given by , so the constitutional supercooling criterion for a binary alloy can be written in terms of the concentration gradient at the interface:
:
The concentration gradient ahead of a planar interface is given by
:
where is the interface velocity, the diffusion coefficient
Diffusivity, mass diffusivity or diffusion coefficient is a proportionality constant between the molar flux due to molecular diffusion and the gradient in the concentration of the species (or the driving force for diffusion). Diffusivity is enco ...
, and and are the compositions of the liquid and solid at the interface, respectively (i.e., ).
For the steady-state growth of a planar interface, the composition of the solid is equal to the nominal alloy composition, , and the partition coefficient
In the physical sciences, a partition coefficient (''P'') or distribution coefficient (''D'') is the ratio of concentrations of a compound in a mixture of two immiscible solvents at equilibrium. This ratio is therefore a comparison of the solub ...
, , can be assumed constant. Therefore, the minimum thermal gradient necessary to create a stable solid front is given by
:
For more information, see Chapter 3 of
In animals
In order to survive extreme low temperatures in certain environments, some animals use the phenomenon of supercooling that allow them to remain unfrozen and avoid cell damage and death. There are many techniques that aid in maintaining a liquid state, such as the production ofantifreeze protein
Antifreeze proteins (AFPs) or ice structuring proteins refer to a class of polypeptides produced by certain animals, plants, fungi and bacteria that permit their survival in temperatures below the freezing point of water. AFPs bind to small ...
s, or AFPs, which bind to ice crystals to prevent water molecules from binding and spreading the growth of ice. The winter flounder
The winter flounder (''Pseudopleuronectes americanus''), also known as the black back, is a right-eyed ("dextral") flatfish of the family Pleuronectidae. It is native to coastal waters of the western north Atlantic coast, from Labrador, Canada to ...
is one such fish that utilizes these proteins to survive in its frigid environment. Noncolligative proteins are secreted by the liver into the bloodstream. Other animals use colligative antifreezes, which increases the concentration of solutes in their bodily fluids, thus lowering their freezing point. Fish that rely on supercooling for survival must also live well below the water surface, because if they came into contact with ice nuclei they would freeze immediately. Animals that undergo supercooling to survive must also remove ice-nucleating agents from their bodies because they act as a starting point for freezing. Supercooling is also a common feature in some insect, reptile, and other ectotherm
An ectotherm (from the Greek () "outside" and () "heat") is an organism in which internal physiological sources of heat are of relatively small or of quite negligible importance in controlling body temperature.Davenport, John. Animal Life ...
species. The potato cyst nematode larva (''Globodera rostochiensis
''Globodera rostochiensis'', commonly known as the golden nematode, golden eelworm or yellow potato cyst nematode, is a plant pathogenic nematode. It is a pest of plants in the family Solanaceae, primarily infesting potatoes and tomatoes, as well ...
'') could survive inside their cysts in a supercooled state to temperatures as low as , even with the cyst encased in ice.
As an animal gets farther and farther below its melting point the chance of spontaneous freezing increases dramatically for its internal fluids, as this is a thermodynamically unstable state. The fluids eventually reach the supercooling point, which is the temperature at which the supercooled solution freezes spontaneously due to being so far below its normal freezing point. Animals unintentionally undergo supercooling and are only able to decrease the odds of freezing once supercooled. Even though supercooling is essential for survival, there are many risks associated with it.
In plants
Plants can also survive extreme cold conditions brought forth during the winter months. Many plant species located in northern climates can acclimate under these cold conditions by supercooling, thus these plants survive temperatures as low as −40 °C. Although this supercooling phenomenon is poorly understood, it has been recognized throughinfrared thermography
Infrared thermography (IRT), thermal video and/or thermal imaging, is a process where a thermal camera captures and creates an image of an object by using infrared radiation emitted from the object in a process, which are examples of infrared ...
. Ice nucleation occurs in certain plant organs and tissues, debatably beginning in the xylem
Xylem is one of the two types of transport tissue in vascular plants, the other being phloem. The basic function of xylem is to transport water from roots to stems and leaves, but it also transports nutrients. The word ''xylem'' is derived from ...
tissue and spreading throughout the rest of the plant. Infrared thermography allows for droplets of water to be visualized as they crystalize in extracellular spaces.
Supercooling inhibits the formation of ice within the tissue by ice nucleation and allows the cells to maintain water in a liquid state and further allows the water within the cell to stay separate from extracellular ice. Cellular barriers such as lignin, suberin
Suberin, cutin and lignins are complex, higher plant epidermis and periderm cell-wall macromolecules, forming a protective barrier. Suberin, a complex polyester biopolymer, is lipophilic, and composed of long chain fatty acids called suberin aci ...
and the cuticle inhibit ice nucleators and force water into the supercooled tissue. The xylem and primary tissue of plants are very susceptible to cold temperatures because of the large proportion of water in the cell. Many boreal hardwood species in northern climates have the ability to prevent ice spreading into the shoots allowing the plant to tolerate the cold. Supercooling has been identified in the evergreen shrubs '' Rhododendron ferrugineum'' and ''Vaccinium vitis-idaea
''Vaccinium vitis-idaea'', the lingonberry, partridgeberry, mountain cranberry or cowberry, is a small evergreen shrub in the heath family Ericaceae, that bears edible fruit. It is native to boreal forest and Arctic tundra throughout the Norther ...
'' as well as ''Abies
Firs (''Abies'') are a genus of 48–56 species of evergreen coniferous trees in the family Pinaceae. They are found on mountains throughout much of North and Central America, Europe, Asia, and North Africa. The genus is most closely related t ...
'', '' Picea'' and ''Larix
Larches are deciduous conifers in the genus ''Larix'', of the family Pinaceae (subfamily Laricoideae). Growing from tall, they are native to much of the cooler temperate northern hemisphere, on lowlands in the north and high on mountains furt ...
'' species. Freezing outside of the cell and within the cell wall does not affect the survival of the plant. However, the extracellular ice may lead to plant dehydration.
In seawater
The presence of salt in seawater affects the freezing point. For that reason, it is possible for seawater to remain in the liquid state at temperatures below melting point. This is "pseudo-supercooling" because the phenomena is the result of freezing point lowering caused by the presence of salt, not supercooling. This condition is most commonly observed in the oceans aroundAntarctica
Antarctica () is Earth's southernmost and least-populated continent. Situated almost entirely south of the Antarctic Circle and surrounded by the Southern Ocean, it contains the geographic South Pole. Antarctica is the fifth-largest contine ...
where melting of the undersides of ice shelves
An ice shelf is a large floating platform of ice that forms where a glacier or ice sheet flows down to a coastline and onto the ocean surface. Ice shelves are only found in Antarctica, Greenland, Northern Canada, and the Russian Arctic. The b ...
at high pressure results in liquid melt-water that can be below the freezing temperature. It is supposed that the water does not immediately refreeze due to a lack of nucleation sites. This provides a challenge to oceanographic instrumentation as ice crystals will readily form on the equipment, potentially affecting the data quality. Ultimately the presence of extremely cold seawater will affect the growth of sea ice
Sea ice arises as seawater freezes. Because ice is less dense than water, it floats on the ocean's surface (as does fresh water ice, which has an even lower density). Sea ice covers about 7% of the Earth's surface and about 12% of the world's oce ...
.
Applications
One commercial application of supercooling is inrefrigeration
The term refrigeration refers to the process of removing heat from an enclosed space or substance for the purpose of lowering the temperature.International Dictionary of Refrigeration, http://dictionary.iifiir.org/search.phpASHRAE Terminology, ht ...
. Freezers can cool drinks to a supercooled level so that when they are opened, they form a slush
Slush, also called slush ice, is a slurry mixture of small ice crystals (e.g., snow) and liquid water.
In the natural environment, slush forms when ice or snow melts or during mixed precipitation. This often mixes with dirt and other polluta ...
. Another example is a product that can supercool the beverage in a conventional freezer. The Coca-Cola Company
The Coca-Cola Company is an American multinational beverage corporation founded in 1892, best known as the producer of Coca-Cola. The Coca-Cola Company also manufactures, sells, and markets other non-alcoholic beverage concentrates and syrups, ...
briefly marketed special vending machine
A vending machine is an automated machine that provides items such as snacks, beverages, cigarettes, and lottery tickets to consumers after cash, a credit card, or other forms of payment are inserted into the machine or otherwise made. The fir ...
s containing Sprite in the UK, and Coke in Singapore, which stored the bottles in a supercooled state so that their content would turn to slush
Slush, also called slush ice, is a slurry mixture of small ice crystals (e.g., snow) and liquid water.
In the natural environment, slush forms when ice or snow melts or during mixed precipitation. This often mixes with dirt and other polluta ...
upon opening.
Supercooling was successfully applied to organ preservation at Massachusetts General Hospital/Harvard Medical School
Harvard Medical School (HMS) is the graduate medical school of Harvard University and is located in the Longwood Medical Area of Boston, Massachusetts. Founded in 1782, HMS is one of the oldest medical schools in the United States and is consi ...
. Liver
The liver is a major Organ (anatomy), organ only found in vertebrates which performs many essential biological functions such as detoxification of the organism, and the Protein biosynthesis, synthesis of proteins and biochemicals necessary for ...
s that were later transplanted into recipient animals were preserved by supercooling for up to 96 hours (4 days), quadrupling the limits of what could be achieved by conventional liver preservation methods. The livers were supercooled to a temperature of –6 °C in a specialized solution that protected against freezing and injury from the cold temperature.
Another potential application is drug delivery. In 2015, researchers crystallized membranes at a specific time. Liquid-encapsulated drugs could be delivered to the site and, with a slight environmental change, the liquid rapidly changes into a crystalline form that releases the drug.
In 2016, a team at Iowa State University
Iowa State University of Science and Technology (Iowa State University, Iowa State, or ISU) is a public land-grant research university in Ames, Iowa. Founded in 1858 as the Iowa Agricultural College and Model Farm, Iowa State became one of the ...
proposed a method for "soldering without heat" by using encapsulated droplets of supercooled liquid metal to repair heat sensitive electronic devices. In 2019, the same team demonstrated the use of undercooled metal to print solid metallic interconnects on surfaces ranging from polar (paper and Jello) to superhydrophobic (rose petals), with all the surfaces being lower modulus than the metal.
Eftekhari et al. proposed an empirical theory explaining that supercooling of ionic liquid crystals can build ordered channels for diffusion for energy storage applications. In this case, the electrolyte has a rigid structure comparable with that of a solid electrolyte, but the diffusion coefficient can be as large as in liquid electrolytes. Supercooling increases the medium viscosity but keeps the directional channels open for diffusion.
In spaceflight
In spaceflight applications, the term is used somewhat differently. Here it refers to cryogenic fuels or oxidizers which are cooled well below their ''boiling'' point (but not below the ''melting'' point). This results in a higher fuel density, and hence a higher capacity of the fuel tanks without increasing their weight. At the same time vaporization losses are reduced.SpaceX
Space Exploration Technologies Corp. (SpaceX) is an American spacecraft manufacturer, launcher, and a satellite communications corporation headquartered in Hawthorne, California. It was founded in 2002 by Elon Musk with the stated goal of ...
's Falcon 9
Falcon 9 is a partially reusable medium lift launch vehicle that can carry cargo and crew into Earth orbit, produced by American aerospace company SpaceX.
The rocket has two stages. The first (booster) stage carries the second stage and payl ...
rocket uses supercooling for its oxidizer.
The term ''superchilling'' is also used for this technique.
See also
*Amorphous solid
In condensed matter physics and materials science, an amorphous solid (or non-crystalline solid, glassy solid) is a solid that lacks the long-range order that is characteristic of a crystal.
Etymology
The term comes from the Greek ''a'' ("wi ...
*Pumpable ice technology
Pumpable ice technology (PIT) uses thin liquids, with the cooling capacity of ice. Pumpable ice is typically a slurry of ice crystals or particles ranging from 5 micrometers to 1 cm in diameter and transported in brine, seawater, food liqui ...
*Subcooling
The term subcooling (also called undercooling) refers to a liquid existing at a temperature below its normal boiling point. For example, water boils at 373 K; at room temperature (293 K) the water is termed "subcooled". A subcooled liqui ...
*Ultracold atom
Ultracold atoms are atoms that are maintained at temperatures close to 0 kelvin (absolute zero), typically below several tens of microkelvin (µK). At these temperatures the atom's quantum-mechanical properties become important.
To reach such low ...
*Viscous liquid
In condensed matter physics and physical chemistry, the terms viscous liquid, supercooled liquid, and glassforming liquid are often used interchangeably to designate liquids that are at the same time highly viscous (see Viscosity of amorphous ma ...
*Freezing rain
Freezing rain is rain maintained at temperatures below freezing by the ambient air mass that causes freezing on contact with surfaces. Unlike a mixture of rain and snow or ice pellets, freezing rain is made entirely of liquid droplets. The rain ...
References
Further reading
* *External links
* * * *Supercooled liquids on arxiv.org
{{Authority control Thermodynamic processes Condensed matter physics Concepts in physics Glass physics